The Struggle for Access to Essential Diabetes Medication
Hundreds of thousands of Australians living with diabetes will continue to pay thousands of dollars annually for life-saving medication that is not included in the Pharmaceutical Benefits Scheme (PBS). This situation has been described as a result of a “broken” process for subsidising medicines, according to the peak industry body.
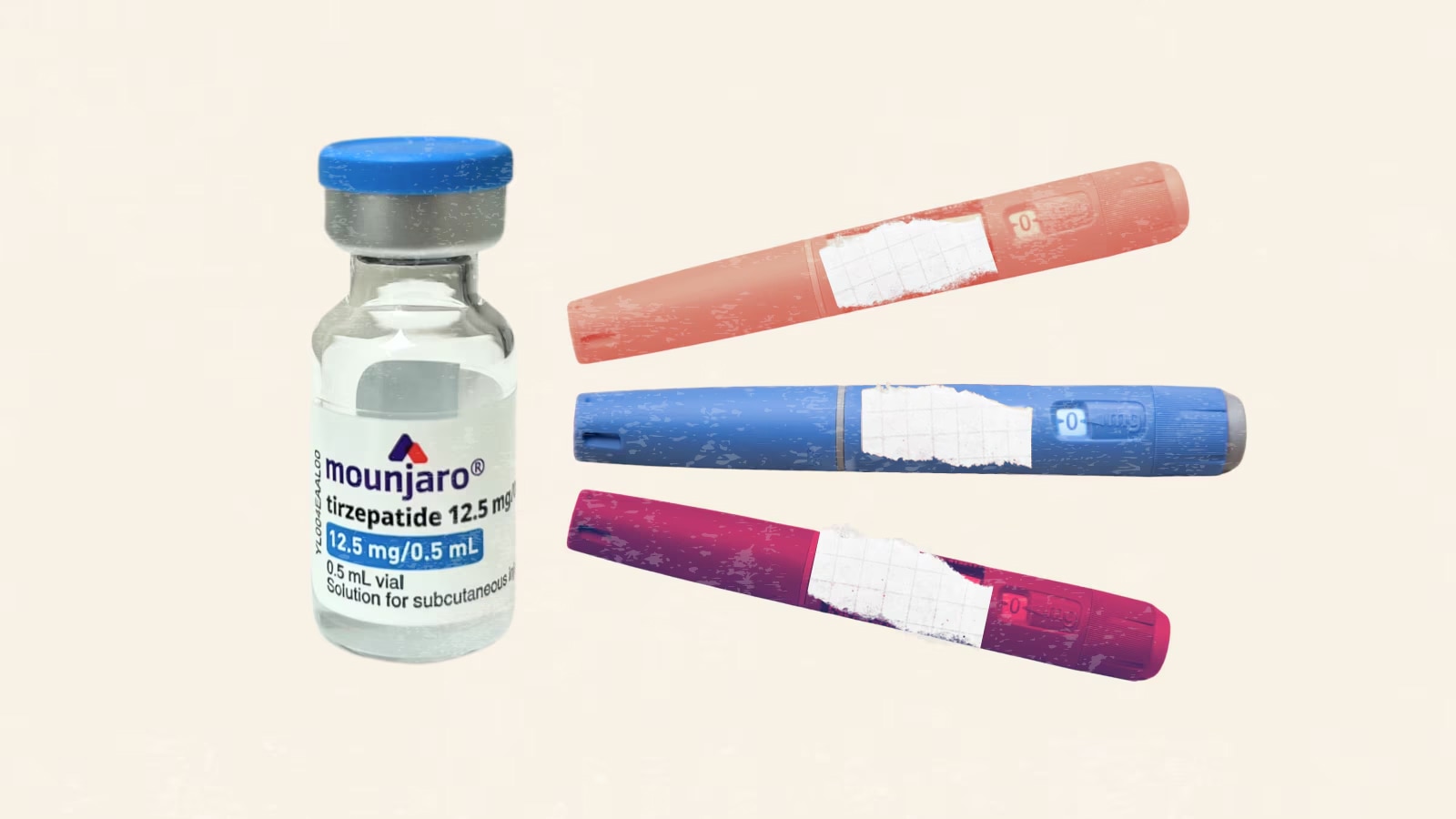
The Pharmaceutical Benefits Advisory Committee (PBAC) had recommended that tirzepatide — an injectable GLP-1 medication marketed as Mounjaro — be subsidised for adults with “inadequately controlled” type 2 diabetes. However, after a prolonged dispute with the federal government, the drug’s manufacturer, Eli Lilly, decided not to proceed with the PBS process. The company cited several concerns, including the price Australia was willing to pay for the drug being too low.
Medicines Australia CEO Liz de Somer expressed disappointment over the outcome, stating that 450,000 Australians with type 2 diabetes could miss out on discounted medication. She highlighted that the system in Australia is outdated and failing to keep up with medical innovation.
The Impact of High Costs on Patients
For patients like Ingrid Baas-Becking, who suffers from both type 1 and type 2 diabetes, the financial burden is immense. She pays nearly $700 every two months for 15 milligrams of Mounjaro, which has significantly improved her health. However, she is forced to ration the drug due to its high cost.
“I’m doing without other things to afford the Mounjaro … I can’t do anything other than buy the Mounjaro,” said Ms. Baas-Becking. She noted that while Mounjaro has helped her lose weight and reduce insulin use, she is only able to take half the dose because of the financial strain.
This situation has led to frustration among patients, advocacy groups, and pharmaceutical companies, who argue that the path to PBS listing is overly complex, involves excessive bureaucracy, and takes too long. The median time for a new medicine to be added to the scheme once approved by the regulator is 22 months.
The Role of Pricing and Negotiations
Keeping PBS prices low comes with trade-offs. Once the PBAC recommends a medication for inclusion, the government negotiates with the pharmaceutical company to strike a commercial agreement that aims to protect taxpayers by mitigating risks, such as uncertainty around the overall cost to the PBS.
Manny Simons, general manager of Lilly Australia and New Zealand, stated that the proposed price conditions were unrealistic and unviable. He argued that the financial risk to the company was disproportionate.
Health economist Philip Clarke from the University of Melbourne’s Centre for Health Policy noted that it is unclear whether Eli Lilly was bluffing to secure a better deal. He pointed out that the actual prices paid by the government for new drugs are often not disclosed, making it difficult to assess the financial implications for the company.
Professor Clarke also mentioned that the government’s commitment to keeping prices low may mean that not every drug will be listed, especially when alternatives are available. He compared this to shopping for cars, where buying every car would not be a good deal.
Government Response and Future Outlook
Health Minister Mark Butler acknowledged the challenges in negotiations with Eli Lilly and stated that his department is reviewing the situation. He emphasized the importance of price negotiations between the government and multinational drug companies in maintaining a successful and sustainable PBS.
Meanwhile, semaglutide, sold under the brand name Ozempic, is already listed on the PBS for people with type 2 diabetes. Wegovy, which contains the same active ingredient, has also been recommended for PBS listing.
Despite these developments, the ongoing issue highlights the need for more urgent action to address the systemic challenges within the PBS. Stakeholders, patients, and their families have long called for reforms to ensure timely access to essential medications. As the debate continues, the focus remains on balancing affordability, innovation, and patient needs.