Major Recall of Nasal Spray Due to Child Safety Concerns
Bayer has issued a significant recall of over 786,000 bottles of its Travel Size Afrin Original Nasal Spray due to concerns about child safety. The products are being recalled because they do not meet the requirements for child-resistant packaging, which could pose serious risks if ingested by young children.
The nasal spray contains imidazoline, a nasal decongestant that is regulated under the Poison Prevention Packaging Act. This law mandates that such products be protected with child-safe packaging to prevent accidental poisoning. If swallowed, imidazoline can lead to severe health issues, including slowed brain activity, difficulty breathing, and heart complications. Children are particularly vulnerable to the effects of this drug, even in small amounts.
According to the Consumer Product Safety Commission (CPSC), the 6 mL nasal spray’s packaging does not meet the necessary child-resistant standards or include the required warning labels. This poses a risk of serious injury or illness from poisoning if the contents are accidentally swallowed by young children.
So far, no injuries or adverse effects have been reported related to this recall. However, the CPSC has emphasized the importance of taking immediate action to ensure the safety of children.
Affected Products and Distribution
The affected products are sold in 6 mL, travel-sized bottles with specific lot numbers: 230361, 240822, 241198, 250066, 250152, 250646, and 250831. These bottles feature the label “Afrin Original Nasal Spray” and “1/5 FL OZ (6 mL)” on the front.

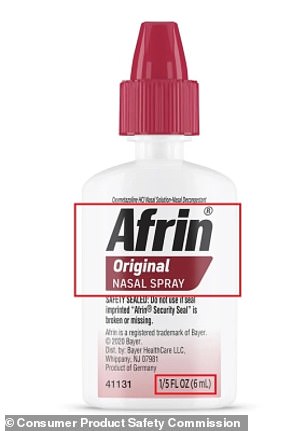

The recalled bottles were available nationwide at convenience stores, airports, and other travel hubs between September 2024 and April 2026. They were typically priced between $7 and $9.
Steps for Consumers
Bayer is urging consumers to take immediate action to secure the recalled bottles out of sight and reach of children. Customers are advised to visit the brand’s website to request a refund. As part of the process, consumers will need to take and submit a photo of the product before disposing of it.
For further assistance, customers can contact Bayer via phone at 800-317-2165, Monday through Friday from 8 am to 8 pm ET. Alternatively, they can use an online recall refund webform.
Understanding the Risks of Imidazoline
Imidazoline is a powerful nasal decongestant that works by constricting blood vessels. It is also found in eye drop brands like Visine and Clear Eyes, where it helps reduce inflammation and redness. While the drug is considered safe when used as directed—either topically in the eyes or inhaled through the nasal passages—swallowing it can lead to rapid toxicity, especially in children.
Symptoms of imidazoline poisoning may include drowsiness, extreme lethargy, low muscle tone, decreased breathing rate, blue lips or fingers, slowed heart rate, low blood pressure, nausea, vomiting, and tremors. Prompt medical attention is crucial if any of these symptoms are observed.
Broader Context of Child-Safe Packaging Issues
This recall follows a similar incident in March, when 27,400 bottles of Tomum Hair Regrowth Treatment containing Minoxidil were recalled due to the same issue of inadequate child-safe packaging. Additionally, over 350,000 bottles of iron-containing dietary supplements were pulled from shelves last month for similar reasons.
In both cases, no injuries or illnesses were reported. However, these recalls highlight the ongoing challenges in ensuring that consumer products, especially those containing potentially harmful substances, are properly secured to prevent accidental ingestion by children.
Consumers are encouraged to remain vigilant and check for any product recalls, particularly when purchasing items that may contain ingredients requiring special handling.






